|
XAFSmass A program for calculations of x-ray absorption for Windows 
Mar 2003 |
|
XAFSmass A program for calculations of x-ray absorption for Windows 
Mar 2003 |
|
pay also your attention to my programs for EXAFS and
XANES
|
|
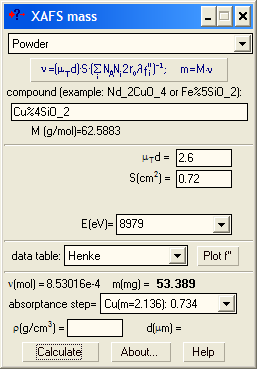
A typical application is the calculation of the mass for a powder sample. The optimal optical sample thickness μd depends on the absorption levels selected for the ionization chambers (see below). Typically, μd is between 2 and 3 (e.g. for a 17.5% absorption level for the 1st chamber and a 50% level for the 2nd chamber, the optimal thickness is 2.41). However, if you get the absorption step more that 1.5, it is recommended to reduce the sample mass to avoid potential thickness effect due to possible inhomogeneity in the wafer. If your sample is diluted and you get a very low absorption step, do not try to make the wafer thicker hoping that you will get better spectra you will not: The optimal thickness gets the best signal-to-noise ratio (it is in this sense it is optimal). You can only try to measure your absorption spectra with another registration technique: in fluorescence or electron yield modes. |
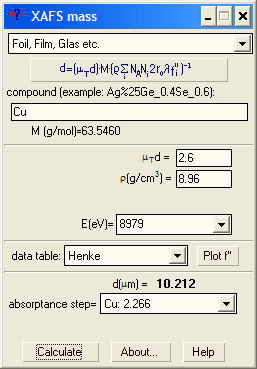
| Here you can calculate the thickness of the sample with known density (usually, a foil). Commercial foils are highly homogeneous in thickness, so that you may ignore large step jumps and pay attention to the total μd only. |
|
|
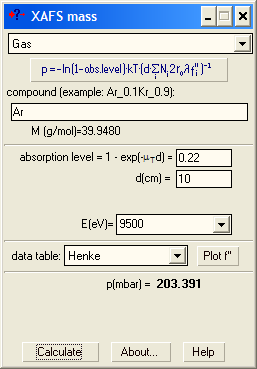
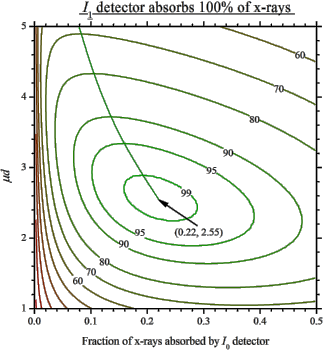
For nitrogen, do not forget the 2: N_2, not just N. Start with the 2nd ionization chamber (IC). If a reference foil is placed between the 2nd and the 3rd IC, the fraction of x-rays absorbed by the 2nd IC is usually set to 50%. If the reference foil is not needed, one can select total absorption (100%). For these two cases the optimal absorption of the 1st IC at a certain μd is found from the following figures showing the levels of signal-to-noise ratio: 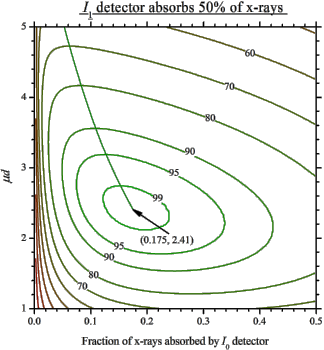
|
|
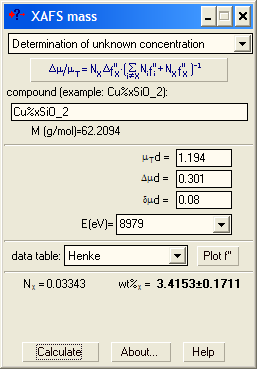
Case 1: You know the composition of the matrix. You need an absorption spectrum taken without the sample (empty spectrum) but with the same state of the ionization chambers. You then subtract it from the spectrum of the sample (e.g. in VIPER) and get a real (i.e. not shifted vertically) absorption coefficient. Determine the value of μd above the edge (μTd), the edge jump (Δμd) and its uncertainty (δμd). Specify the chemical formula with x. |
|
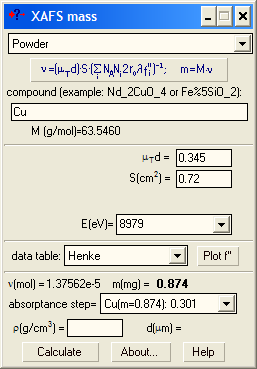
Case 2: You know the sample mass and area. Determine the edge jump (Δμd). For the pure element find such a value for μTd that the absorption step in the pull-down list was equal to your experimental Δμd. This will give you the mass of the element of interest. Just divide it by the total mass to get the weight percentage. |
|
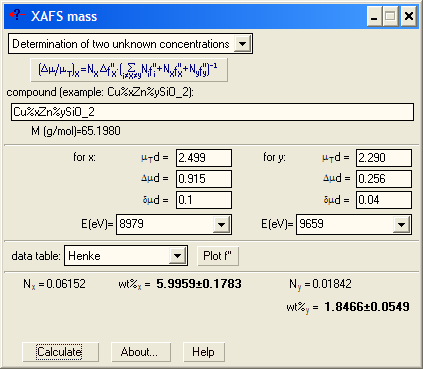
| Here you also need empty spectra (for each of the two edges) to find the unshifted values for absorption coefficient above both edges. |
|
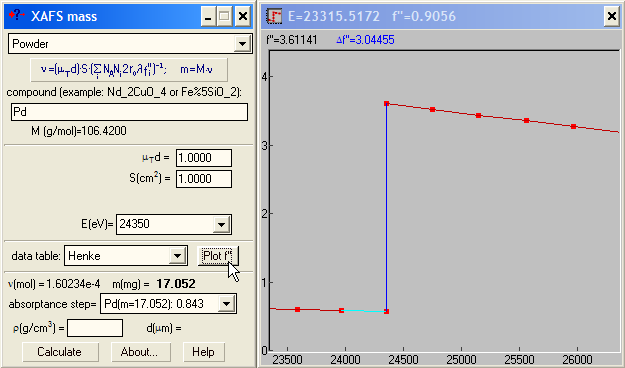
|
If you need to know the scattering factor f'' at different energies and/or its jump at an edge (Δf''), XAFSmass provides a graphical tool for this. For example, you may need these values to determine the composition of a binary compound if you have the experimental edge heights at two edges. The absorption step Δμd at the absorption edge of energy E is proportional to Δf''ν/E, where ν is the amount of (resonantly) absorbing atoms in mole. Hence, the atomic ratio of two elements in the same sample is νA/νB = (Δμd)A/(Δμd)B ·[Δf''A/Δf''B · EA/EB]. For binary compounds AxB1-x the concentration x is calculated then as x = (νA/νB)/[1+(νA/νB )]. |
|
pay also your attention to my programs for EXAFS and
XANES
|