URL: https://www.desy.de/news/news_search/index_eng.html
Breadcrumb Navigation
DESY News: Scientists observe unusual melting of diamond
News
News from the DESY research centre
Scientists observe unusual melting of diamond
An international team of scientists has found evidence of an unconventional melting process in diamond induced by an X-ray laser beam. While in conventional melting the atoms of a sample start moving stronger and stronger until their bonds break due to heating, the incredibly intense X-ray laser flashes ripped the bonds apart straight away, and only afterwards the atoms started moving due to heating. The scientists around Ichiro Inoue from the RIKEN SPring-8 Center in Japan, Eiji Nishibori from University of Tsukuba in Japan and DESY scientist Beata Ziaja report their observations in the journal Physical Review Letters.

XFELs generate X-ray flashes (called pulses) with femtosecond (quadrillionths of a second) durations and a peak brilliance of more than a billion times that of X-ray tubes. When the XFEL pulse is focused to micrometer- or nanometer-scale spots on a sample, many electrons are excited at once, causing irreversible structural changes. Understanding how matter responds to these intense X-ray pulses is essential for all applications. Now, the team has visualised the XFEL-matter interaction processes by using XFEL beams from the SPring-8 Angstrom Compact free electron LAser (SACLA).
The researchers used diamond as a sample and introduced a “pump-probe” technique using twin XFEL pulses: “The first pulse (pump pulse) excites the diamond and the second pulse (probe pulse) with a controlled delay time was used to investigate the structure of the sample,” explains Inoue. By carefully analysing the diffraction intensity of the probe pulse, the researchers determined the spatial electron density in diamond after the excitation with the pump pulse.
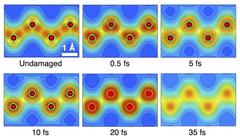
The measured results were interpreted on the basis of dedicated theoretical simulations provided by a team including DESY members and international collaborators. These simulations showed that the observed displacement of carbon atoms was due to an ultrafast transition of the diamond's crystal structure caused just by the presence of many electrons excited by the intense X-ray FEL pulse. The structure transition forced atoms to quickly relocate their positions. “This femtosecond transition is called 'non-thermal' as it is not triggered by a much-longer-taking, 'thermal' heating of atoms in the crystal lattice,” explains Ziaja. “The simulations allowed to unambiguously identify the mechanism and the stages of the observed transition which ended in rapid diamond melting.”
Additional theory considerations confirmed the correct interpretation of the information contained in the so-called diffraction patterns that the X-ray pulses leave on the detector after being scattered by the diamond's crystal lattice. This supported the conclusion on the observed structural transition.
“The X-ray-induced non-thermal melting should be ubiquitous for many experiments with high-intensity XFEL pulses,” comments Nishibori. “In particular, our finding can make a huge impact on developing methodologies for structure determination with high-intensity XFEL pulses, as in this intensity regime, the X-ray-induced damage occurring during the irradiation cannot be neglected.”
Reference:
Atomic-scale visualization of ultrafast bond breaking in x-ray-excited diamond; Ichiro Inoue, Yuka Deguchi, Beata Ziaja, Taito Osaka, Malik M. Abdullah, Zoltan Jurek, Nikita Medvedev, Victor Tkachenko, Yuichi Inubushi, Hidetaka Kasai, Kenji Tamasaku, Eiji Nishibori, and Makina Yabashi; Physical Review Letters, 2021; DOI: 10.1103/PhysRevLett.126.117403



